|
4/19/2024 0 Comments Who created the atomic theory This constant rate of decay can be used as a ‘clock’ to gauge the age of such things as rocks, fossils etc. He observed that any quantity of radioactive isotope takes the same amount of time for half of it to decay. Loss of an alpha particle means a lowering of the mass number by 4 and atomic number by 2. Contrary to Dalton’s view of the atom, he demonstrated that an atom can be ‘destroyed’. For his contribution, together with his investigation into the chemistry of radioactive substances, Rutherford received the Nobel Prize in Chemistry in 1908. Soddy collaborated on the theory of radioactive disintegration and the transmutation (rearrangement) of nuclei and atoms. Using similar techniques to Thomson, he showed that α-rays are helium ions (He 2+) whilst β-rays are, like cathode rays, electrons. He studied two of the three types of radiation emitted by uranium which he named α-rays and β-rays. He was the central figure in the study of radioactivity. Thomson, and possibly the greatest experimentalist since Faraday. He was awarded the Nobel Prize in Chemistry, in 1922. Aston would go on to use their electro-magnetic focusing technique to identify 212 of the 254 naturally occurring, stable isotopes. Thomson, identified two isotopic forms of neon. The chemical properties of elements are defined by the properties of orbital electrons, whilst radioactivity is determined by the nature of the nucleus. 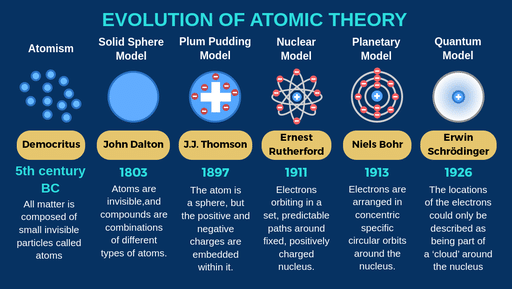
In microchips, electrons are shuffled around in a way prescribed by computer code. 
Thomson, Bell and others utilised them to bring about the electric telegraph, telephones, electric generators and motors, and the electronics industry-encapsulating smart phones and computers. Background scientists such as Davy and Faraday exploited their loose boundedness to produce electricity. Electrons are the most useful of the sub-atomic particles because of their detachability. This fundamental particle was later given the name “electron”. Thomson identified a universal constituent of matter and effectively overturned Dalton’s indivisibility hypothesis. In an evacuated glass vessel, he produced cathode rays-negatively charged particles in transverse motion. Thomson was awarded the Nobel Prize in Physics, in 1906, for his theoretical and experimental investigations into the conduction of electricity by gases. Thomson succeeded his tutor, Lord Rayleigh, as Cavendish Professor of Experimental Physics at the University of Cambridge. According to his law of multiple proportions, in a chemical reaction, atoms combine in small whole number ratios. A chemical reaction is simply a rearrangement of atoms. 
All atoms of one element are identical to each other but different from other elements. In 1803, he asserted that all matter, whether gas, liquid or solid, is composed of small, indivisible particles. John Dalton found that the total pressure created by a mixture of gases is the sum of their partial pressures. Thomson, Rutherford and Chadwick) put forward an increasingly sophisticated architectural model of matter. Between 18, four British scientists (viz Dalton, J.J. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction.For over two millennia, philosophers and scientists had theorized about the composition of matter. Scientists did not account for the gases that play a critical role in this reaction. \) shows that the burning of word does follow the law of conservation of mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
